Student-directed learning
can be messy, noisy and
seemingly out-of-control at times,
but I have learned to
embrace some degree of chaos
as it is often a sign of true learning
can be messy, noisy and
seemingly out-of-control at times,
but I have learned to
embrace some degree of chaos
as it is often a sign of true learning
Reflection - Constructivism
Portfolio question: Subject Matter and Pedagogical Knowledge
|
Constructivist teaching and learning is characterized by learners who are active in constructing their own knowledge through social interaction.
The Human Periodic Table Lesson is one lesson I did which exemplifies how I used a constructivist approach to help students learn about the elements, their atomic structure and their organization within the periodic table. The enduring understanding for this lesson is that the characteristics of the elements - starting from the one with the lowest atomic number to the highest - follow a recurring pattern of physical traits and chemical reactivity. For example, elements number 2, 10 and 18 (Helium, Neon and Argon respectively) are all gases that are non-reactive. This periodicity is the basis by which the elements are organized in Mendeleev's Periodic Table. It is furthermore determined by the number of electrons in the outermost orbital of the atom (valence) which in turn is dictated by the atomic number and the rules restricting how electrons are organized within the atom. |
|
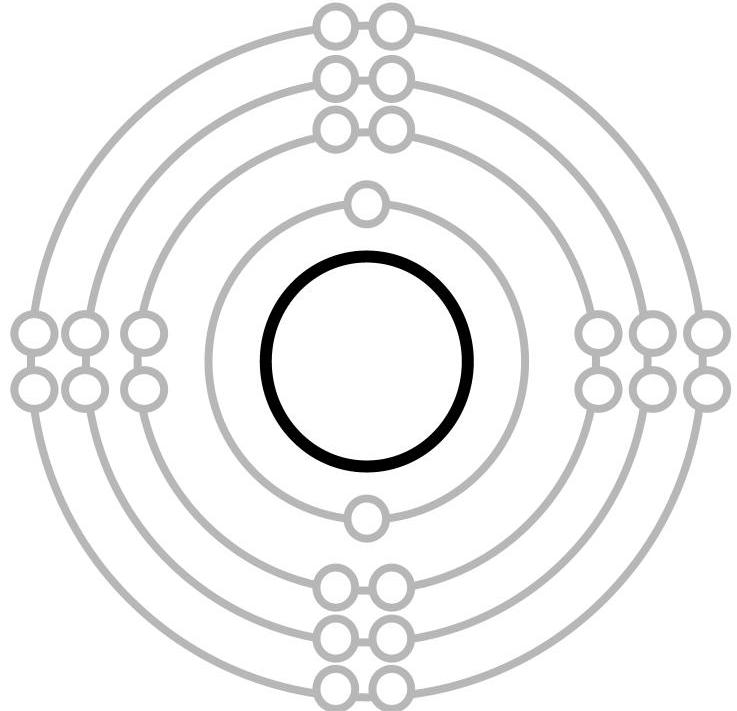
We began the lesson by discussing these rules using the simpler Bohr representation of the atom with electron orbitals. The first orbital closest to the centre of the atom holds a maximum of two electrons, whereas the next 3 orbitals hold a maximum of 8 (Figure 1).
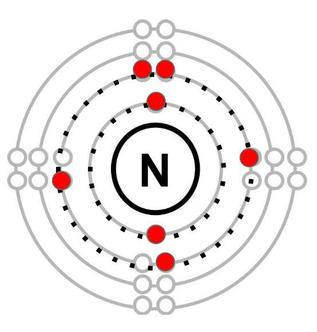
Orbitals are filled with electrons starting from the one closest to the centre. The first two electrons of nitrogen (7 electrons) go into the first orbital. To help students understand how the third to seventh electrons of nitrogen (atomic number 7) are assigned to the second orbital, I used an analogy of siblings sharing bedrooms. In the second orbital, there are 4 “bedrooms”, each with two “beds”, for a total of eight spots that must be shared among the five remaining siblings (or electrons). Since each sibling would prefer to have a room of their own, they will naturally fill these orbitals so that the maximum number of electrons do not have to share. This would mean then that in nitrogen, three electrons get their own room and only two have to share (Figure 2). After this brief instruction, students were given the opportunity to apply their understanding of this rule to an element I had assigned them and to verify their answers with one another. After they had created the Bohr diagram of their element, they were prompted to determine the number of valence electrons it had (the number of electrons in its outermost orbital). |
|
The culminating task was to come up with several logical arrangements of the first twenty elements using the information they had – atomic number and number of valence electrons. We did this activity as a large group in the hallway where the students could move freely about and discuss the various organization options (Figure 3). Having these learners argue through the reasoning among themselves was an important part of constructing their understanding of the elements. Afterwards, we compared their results to Mendeleev’s Periodic Table.
Constructivist learning theory supposes that the students, having been involved in the process of coming up with a very similar arrangement themselves, would better appreciate and remember the organization of the Periodic Table than if had they been simply told how it was arranged. |
Learnings
At first I was afraid to give up control of the class to the activity, but my associate teacher, who knew the class well, encouraged me to do so. There were moments when I felt unsure and wanted to wrap up the activity, as it appeared very disorganized, but with some time and faith, the students were able to successfully put together several reasonable and logical arrangements.
Initially I did not consider how the duplication of certain atomic numbers (due to the complicated nature of higher atomic numbers I did not go above twenty) might affect the activity by creating a group of students who were not sure when to be involved and who at times may have felt left out. Now that I see these implications I have ideas (under goals) of how to better involve everyone.
While it is a great activity for the kinesthetic and visual learners, a major question this experience raised for me was whether this activity was worth the time in terms of what the students learned from it. As with all decisions, many factors need to be considered such as, whether time and space are available and whether the students have already had many kinesthetic activities or seem restless and in need of some movement.
Initially I did not consider how the duplication of certain atomic numbers (due to the complicated nature of higher atomic numbers I did not go above twenty) might affect the activity by creating a group of students who were not sure when to be involved and who at times may have felt left out. Now that I see these implications I have ideas (under goals) of how to better involve everyone.
While it is a great activity for the kinesthetic and visual learners, a major question this experience raised for me was whether this activity was worth the time in terms of what the students learned from it. As with all decisions, many factors need to be considered such as, whether time and space are available and whether the students have already had many kinesthetic activities or seem restless and in need of some movement.
Goals |
Some revisions I plan to make in the future include ensuring that there are modifications for students who have less experience with self-direction. The activity could be made simpler and less open-ended by having the class discuss the possibilities in class before the activity or even giving them specific arrangements to complete. Another modification to help reduce the difficulty of the activity would be to divide the class into two groups and have them do the activity separately. The smaller numbers would make it easier for the students to organize themselves, and give more students a chance to play leadership roles. Additional layers of organization could be added by assigning roles, or having the students come up with roles and deciding among themselves who will assume these roles. |
Next time my strategy will be to also have alternate individual activities so that students who do not enjoy working in groups can have something to do. An example would be to have element cutouts for them to arrange themselves on a worksheet.
In future, one could explore how this could be extended to an enriched class such as doing a Lewis-Dot diagram version using valence electrons only and assigning atomic numbers higher than 20 (which cannot be done with Bohr diagrams).